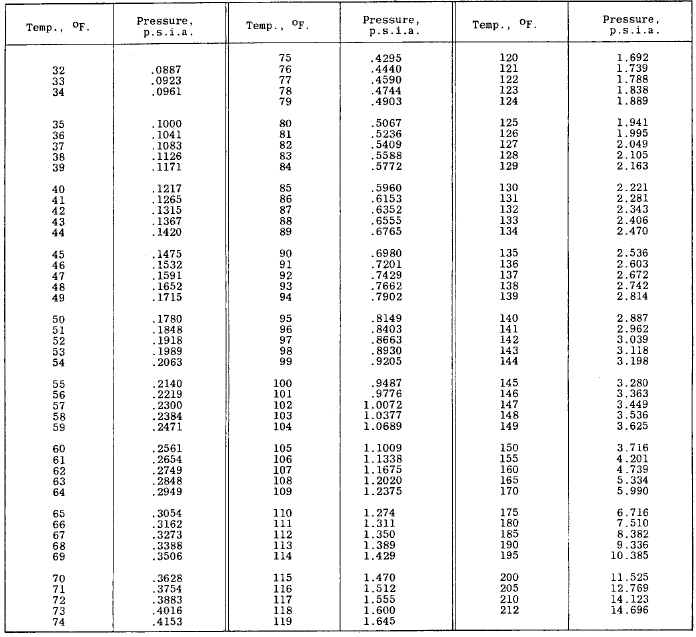
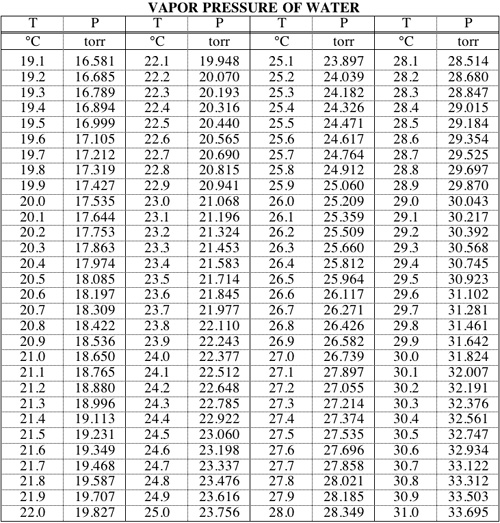
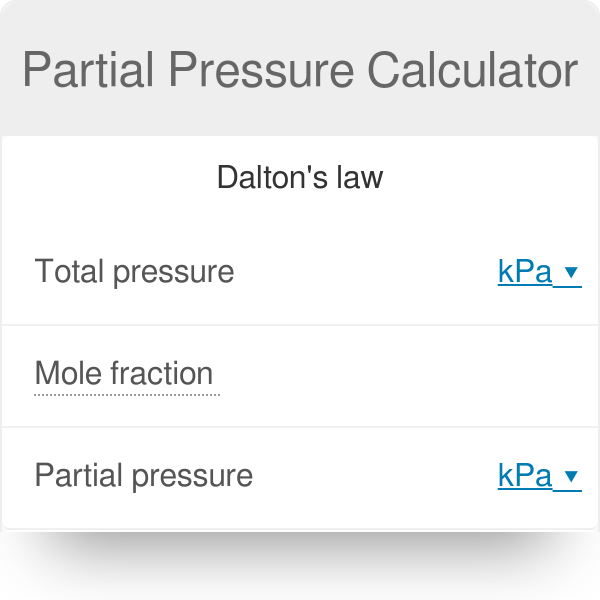
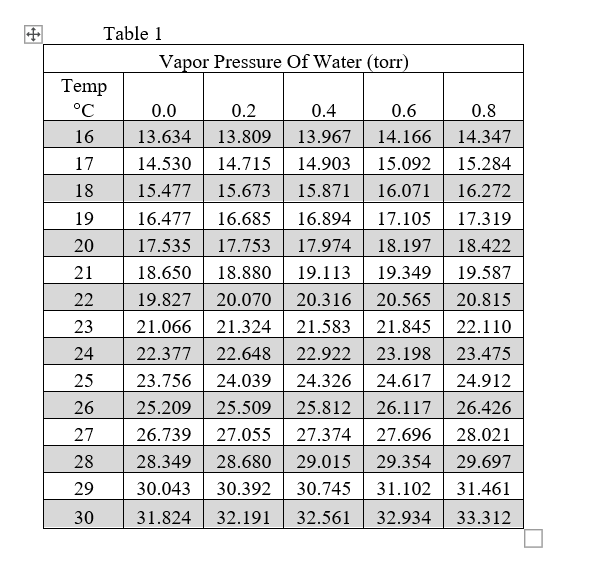
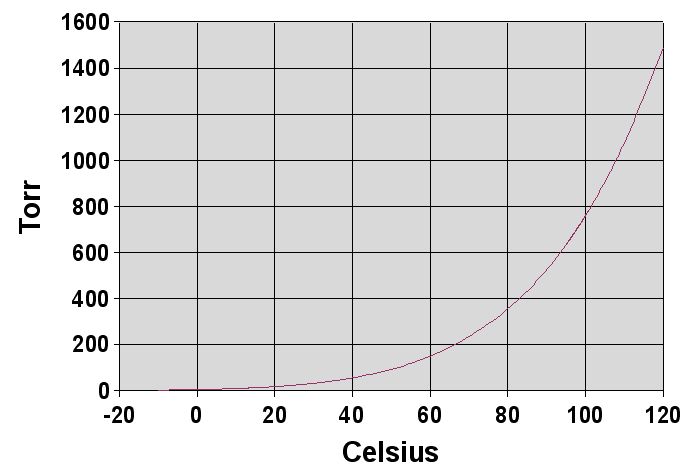
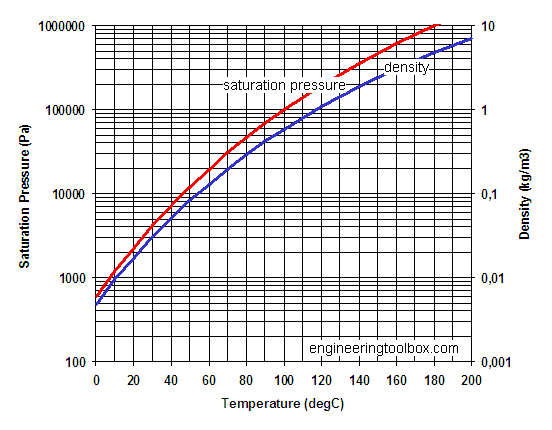
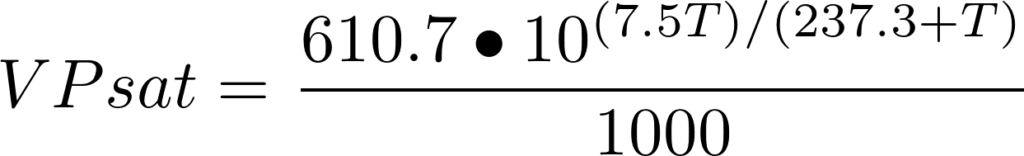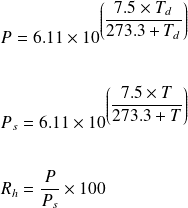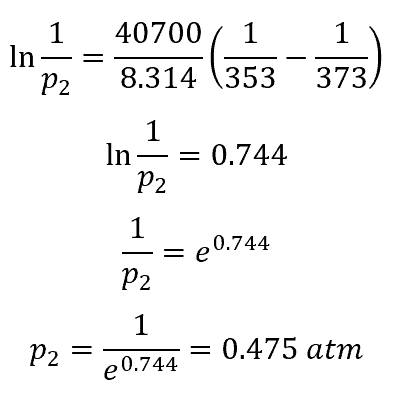A solution contains 500 g of LiCl (MM=42g/mol) in 1.5 L of water. What is the vapor pressure of water (in torr) over the solution at 25.0 oC? (The vapor pressure of

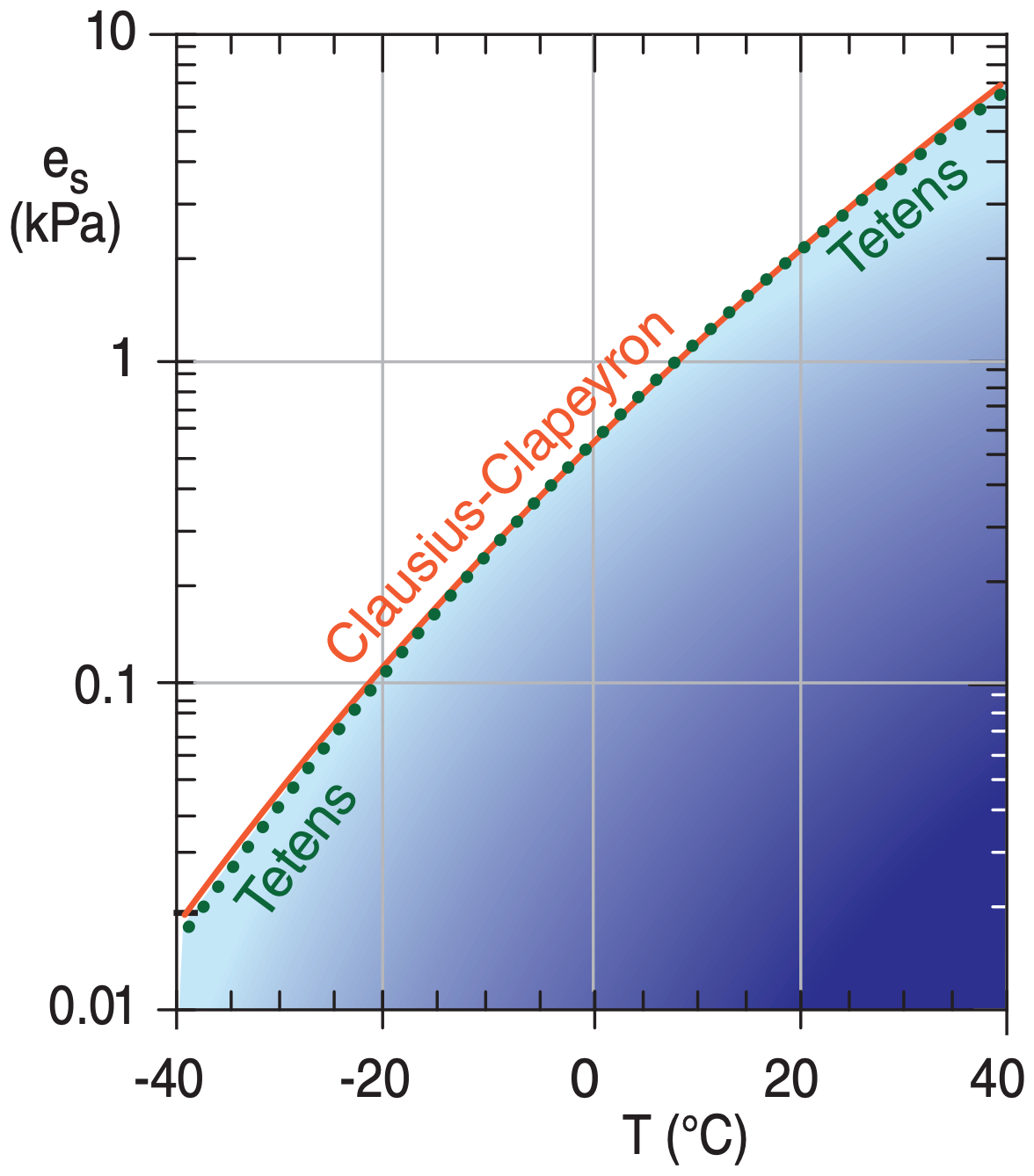
SOLVED: Over a water surface, the saturation vapor pressure (es) is related to the air temperature (T) by the following equation: 17.27T es = 611exp(237.3 + T) where es is in pascals
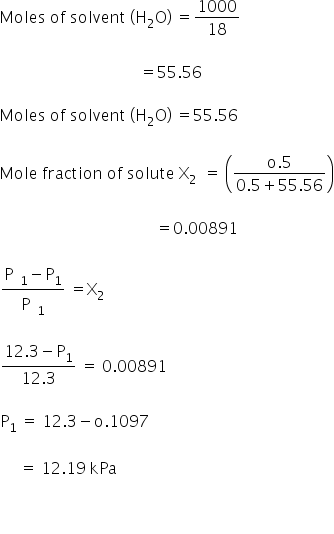
The vapor pressure of water is 12300 Pa at 27 C. Calculate the vapor pressure of 0.5 M solution of a non dissociating/non associating solute in it. - hk3hf2ll
Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.