SOLVED: Calculate percent recovery for each dye value: extract using the grape Kool-Aid spectrum as the starting point. Generally, percent recovery is calculated using this formula: % recovery = (recovered amount /

SOLVED: Calculate Percent Recovery: recovered mass of caffeine (g) % recovery × 100% = initial mass of caffeine (g)

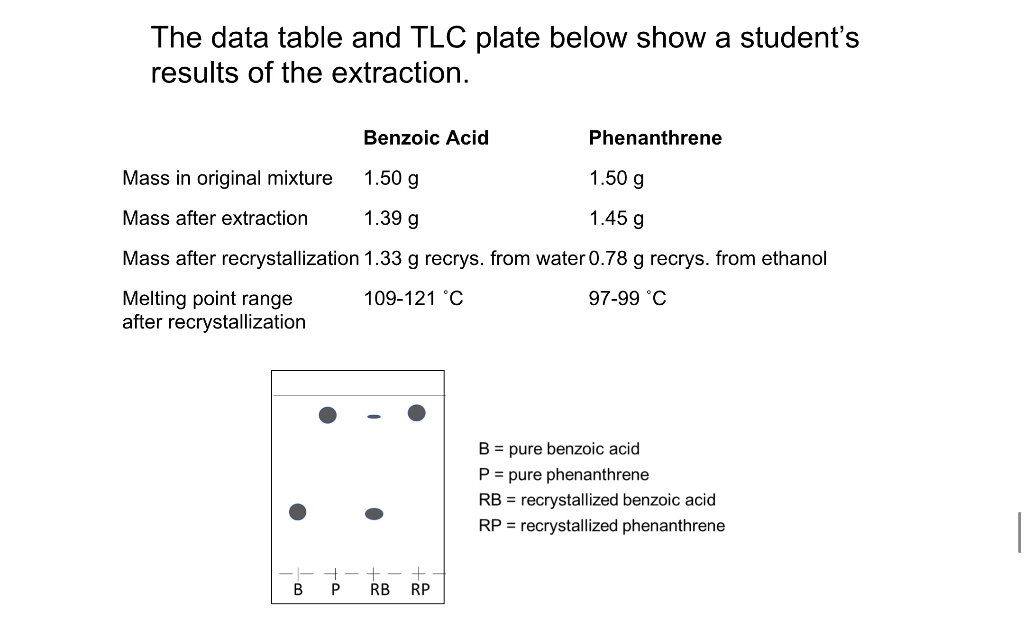
SOLVED: Analysis of product: 1: Calculate the percent recovery based on the amount of tea originally used and a final mass of 0.20g of caffeine obtained. (Amount of tea used was 21.10g)

SOLVED: Calculate the percentage recovery for each product. Note that these calculations might also be referred to as "experimental percent composition calculations" and are calculated by dividing the mass of each product

Percent Yield and Percent Error Calculations | Science, Chemistry, Percent Yield, percent error | ShowMe







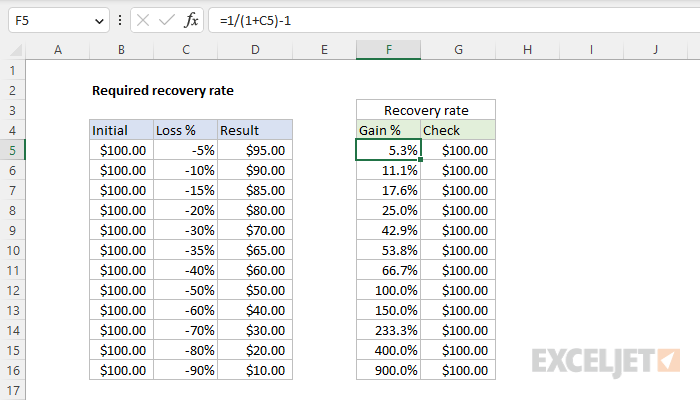
:max_bytes(150000):strip_icc()/recovery-rate.asp_Final-a8fac2c32d704c628b7408edd9604684.png)