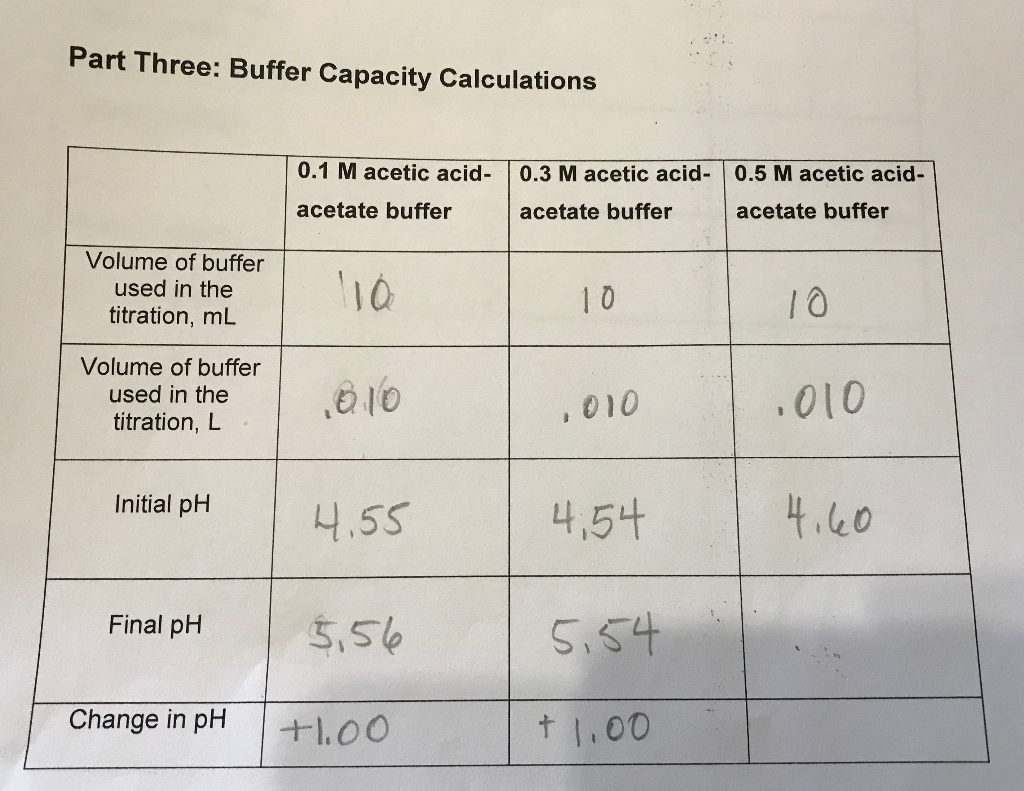
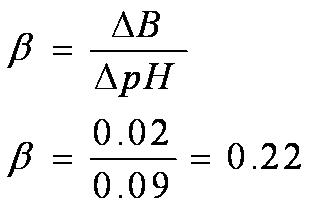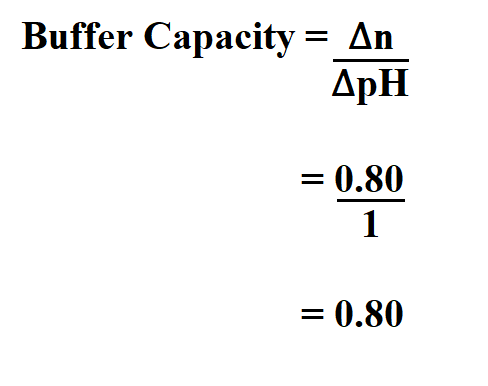SOLVED: Using eq: 7: 2.3 BC = 1 1 + WA CB calculate the buffer capacity (BC) of your buffer solution before and after dilution with water; and enter the values obtained

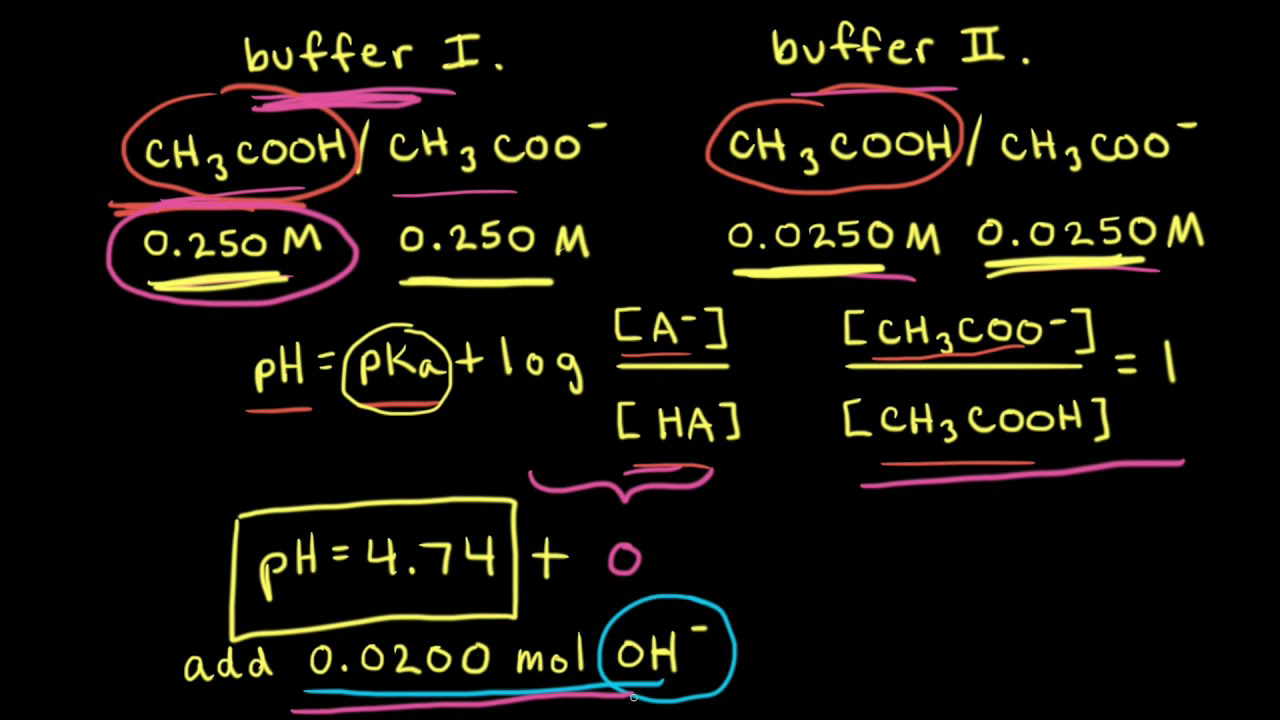
pH calculations and more in fundamentals of pharmaceutics. : How to calculate exact buffer capacity?

OneClass: Buffer capacity is a measure of a buffer solution's resistance to changes in pH as strong a...
4. 0.40 g of NaOH was added to 1 litre of a buffer solution. Its buffer capacity is found to be 0.06. Its pH change will be ? 0.33 0.17 1.0 0.01

Table 2 from Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar