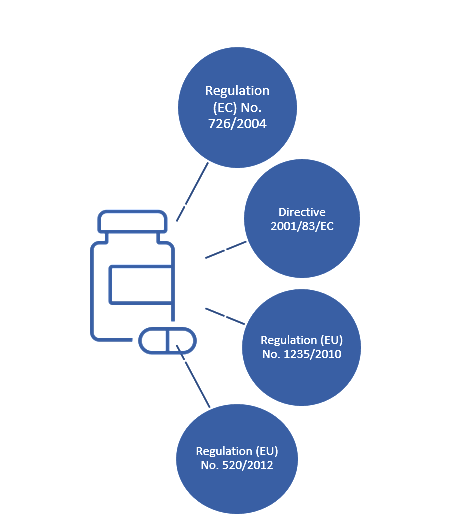
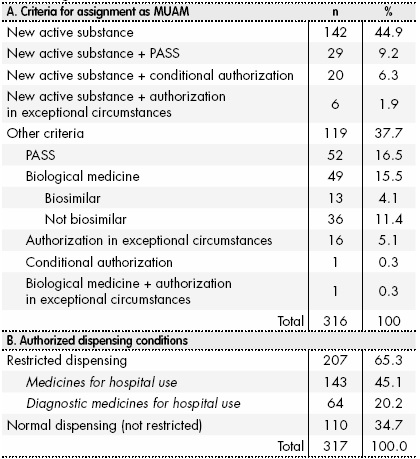
EU Commission Report on National and EMA experience on Medicines subject to Additional Monitoring released today

PDF) Does additional monitoring status increase the reporting of adverse drug reaction s ? An interrupted time series analysis of EudraVigilance data

Promoting and Protecting Public Health: How the European Union Pharmacovigilance System Works. - Abstract - Europe PMC

Improving the Safety of Medicines in the European Union: From Signals to Action - Potts - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library

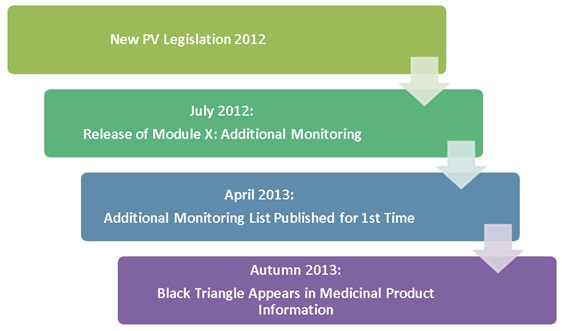
News from the EACPT: New European Medicines Agency advice on black triangle prompts for medicines monitoring
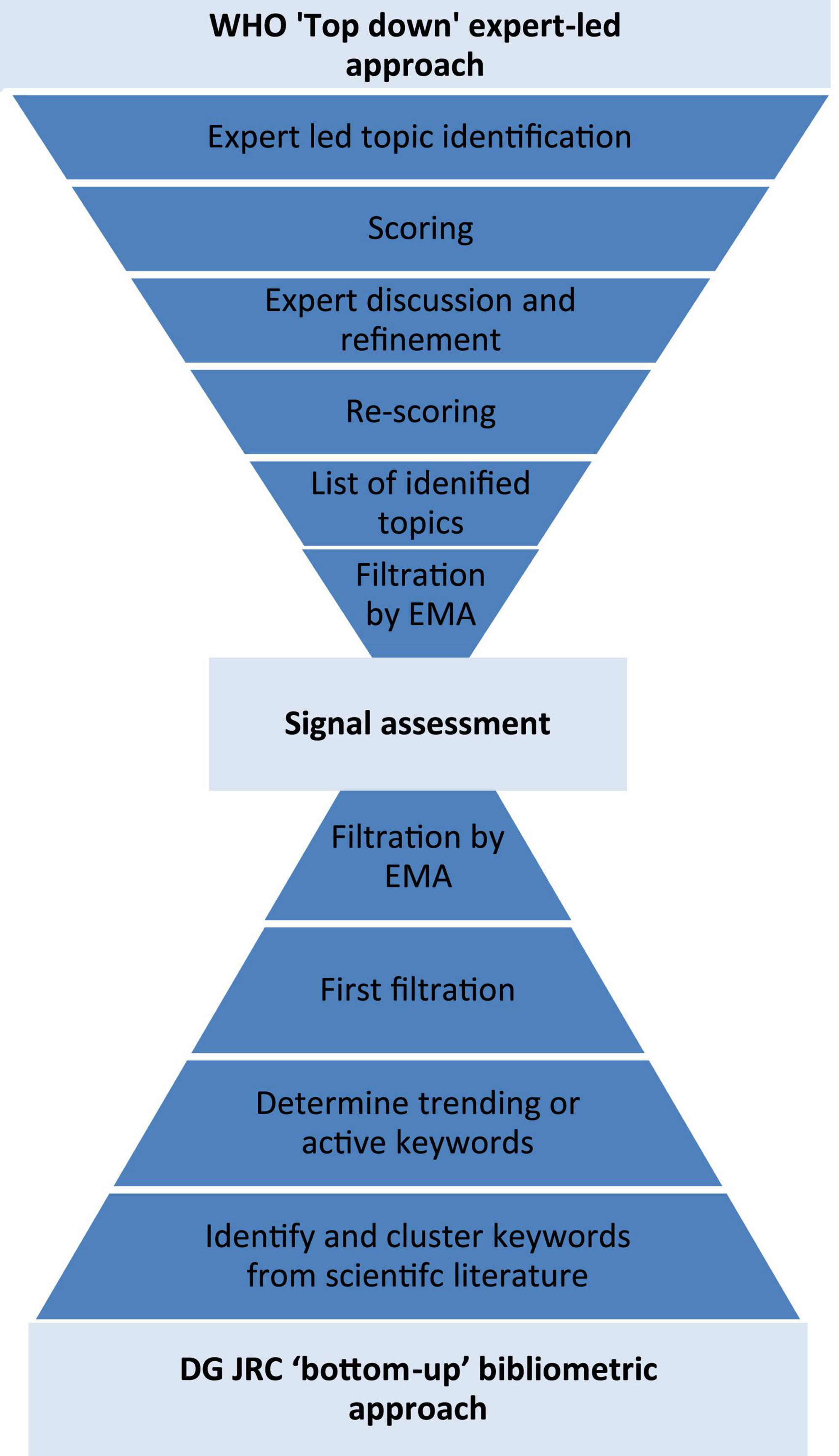
Frontiers | Health horizons: Future trends and technologies from the European Medicines Agency's horizon scanning collaborations